What is Antimicrobial Resistance (AMR)?
If we do not address antimicrobial resistance (AMR) now, we will lose the ability to safely perform routine medical procedures and surgeries. Antimicrobial resistance occurs when bacteria, viruses, fungi, and parasites mutate naturally over time and no longer respond to modern medicines making infections harder to treat. As a result, standard antibiotic treatments become ineffective and infections persist, increasing the risk of severe consequences to the patient.
How does Antibiotic Resistance Occur?
The development of resistant bacteria is a natural phenomenon that is exacerbated and accelerated by the inappropriate use of antibiotics in the fields of human and animal health and the inadequate infection control.
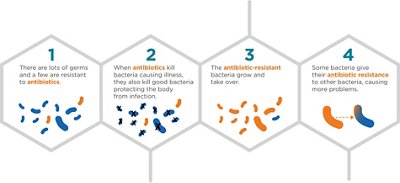
1Source: CDC
What is the Cost of Antimicrobial Resistance?

The cost of antimicrobial resistance (AMR) is significant and can be measured in terms of human lives lost. It’s a complex issue that requires global attention and coordinated efforts to address.

According to data from a 2019 study in The Lancet2
What is the Solution to Fight the AMR Crisis?
Antimicrobial Stewardship (AMS) is a commitment to preserve antibiotic efficacy for future generations through appropriate use of antimicrobial drugs. The goal of antimicrobial stewardship initiatives is to ensure that the right antibiotic is given to the right patient at the right time, with the right dose and via the right route – causing the least harm to the patient. In realistic terms, it is a multidisciplinary approach that aims to ensure that patients benefit from the most effective antibiotic treatments, while limiting the side effects and costs of unnecessary treatments.
AMS programs help fight antimicrobial resistance by providing a framework for responsible antibiotics prescribing and use, optimizing individual therapy, helping reduce the spread of resistance and raising awareness & education.
- Improve Labs: countries need medical labs to identify bacteria and choose the right drugs to treat them
- Collect & Share Data: countries need systems to track cases and report results globally to make better policy decisions
- Use Antibiotics Wisely: to ensure antibiotics are here when we need them, they must be prescribed and taken correctly now
- Take Measures to Prevent Infections: especially in healthcare settings, effective infection control practices are critical
What is the bioMérieux Solution for AMR/AMS?
bioMérieux is your trusted partner in today’s healthcare systems. We provide the most comprehensive diagnostics and expertise, enabling hospital laboratories to provide actionable evidence and insights to clinicians, partnering together on improving patient outcomes and creating more effective stewardship programs to fight AMR.
Our complete AMS solution encompasses more than just traditional state-of-the-art lab technology. Because at bioMérieux we fully understand your antimicrobial stewardship (AMS) needs, our offer translates into 4 key areas where we help hospitals and labs worldwide evolve to suit today’s needs and tomorrow’s concerns:
- Diagnostic Solutions: Diagnostic tools help clinicians determine timely and appropriate treatment which help to reduce the spread of antimicrobial resistant infections. As the largest in vitro diagnostics company committed to antimicrobial stewardship, we help maximize the use of diagnostic solutions to detect, identify, and treat today’s most urgent drug-resistant pathogens.
- Advanced Analytics: We support healthcare systems in their efforts to deliver evidence-based clinical care with our analytic platforms that help to optimize efficiency and increase collaborative communication.
- Services: Improve lab efficiency and accelerate the medical and economic value of stewardship.
- Educational Modules: Educate hospital stakeholders to ensure antimicrobial stewardship programs’ success.
Disclaimer: Product availability varies by country. Please consult your local bioMérieux representative for product availability in your country.
Our Antimicrobial Stewardship Offer
Put Diagnostics at the Heart of Antimicrobial Stewardship to Drive Informed Therapy Decisions
bioMérieux has the most comprehensive solutions on the market all along the patient pathway from antibiotic initiation to optimization and discontinuation. Comprehensive solutions exist today to slow the spread of antimicrobial resistance and accelerate your stewardship program success. bioMérieux is dedicated to helping you as a healthcare professional access actionable results faster because time-to-diagnose and therapeutic efficacy are critical.
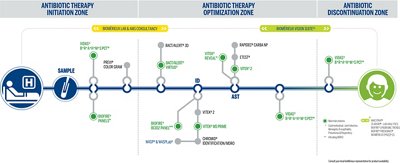
Metromap
AMS Solution
bioMérieux can support you with our solutions that confirm or rule out bacterial infection and identify the causative pathogen for informed patient management and avoiding unnecessary antibiotic use.
-
VIDAS® B•R•A•H•M•S PCT™
Important Biomarker for Improved Patient Management
Detects procalcitonin, a biomarker that aids in the risk assessment for progression to severe sepsis and septic shock. PCT also aids in decision making on antibiotic therapy for patients with lower respiratory tract infections (LRTI).
-
BIOFIRE® Respiratory 2.1 (RP2.1) Panel & Respiratory 2.1 plus (RP2.1plus) Panel
1 Test. Up to 23 Targets. ~45 Minutes.
The BIOFIRE RP2.1 and RP2.1plus Panels use the syndromic approach to accurately detect and identify the pathogens most associated with respiratory infections. Fast and comprehensive results may enable better-informed diagnosis and treatment of patients.
-
PREVI® COLOR GRAM
A Market-Leading Automated Gram Stainer Offering Confidence in Results
Gram staining is a key step in your microbiology workflow, and PREVI® COLOR GRAM is the system to streamline it and provide timely staining results for impactful decisions.
To optimize antibiotic therapy guidance and improve patient outcomes faster, our solutions help identify pathogens and determine their resistance profiles in order to select the most appropriate treatment.
-
BACT/ALERT® VIRTUO®
Automated System for Faster Blood Culture
The first fully automated blood culture system, helping to refine and redesign laboratory workflow to maximize efficiency, while providing faster clinically impactful results.
-
BACT/ALERT® 3D
Accurate Blood Culture Results
BACT/ALERT® 3D brings flexible and modular automated detection systems for any size laboratory that needs to meet blood culture guidelines efficiently, economically, and effectively.
-
VITEK® MS PRIME
New-Generation Mass Spectrometry Microbial Identification System
VITEK® MS PRIME combines microbiology expertise with innovation taking mass spectrometry to the next level by maximizing the impact of daily laboratory workflow for better patient care.
-
BIOFIRE® FILMARRAY® TORCH
Easier testing. Faster results.
Syndromic infectious disease testing with BIOFIRE® FILMARRAY® TORCH is the fastest way to better results.
-
VITEK® 2
Fully integrated Identification and Antimicrobial Susceptibility Testing
VITEK® 2, the leading automated system for routine antimicrobial susceptibility testing, provides efficient workflow, faster AST results. Its fully-integrated ID & AST approach ensures superior performance to rapidly and confidently guide therapy.
-
VITEK® REVEAL™
Fast Antimicrobial Susceptibility Testing (AST) for Bloodstream Infections
The newest addition to our Sepsis Solution, VITEK® REVEAL™ offers fast AST results to effectively and efficiently manage bloodstream infections.
-
WASP®
Automated Microbiology Specimen Processing Instrument
Quality diagnostic results depend on high standard specimen processing. WASP® is the smart solution for truly comprehensive automation – taking you far beyond plating and streaking to address all aspects of microbiology specimen processing.
WASP® System is manufactured by COPAN Company. -
WASPLab®
Integrated Lab Optimization and Automation
The flexible automated specimen processing, incubation, and reading solution that adapts to your unique lab setup and workflow – and even grows with you over time.
WASPLab® System is manufactured by COPAN Company. -
VIDAS® B•R•A•H•M•S PCT™
Important Biomarker for Improved Patient Management
Detects procalcitonin, a biomarker that aids in the risk assessment for progression to severe sepsis and septic shock. PCT also aids in decision making on antibiotic therapy for patients with lower respiratory tract infections (LRTI).
To help answer when the patient can be safely discontinued from antibiotic treatment, our solutions help monitor the patient's response to personalized treatment and discontinue antibiotics as early as possible.
-
VIDAS® B•R•A•H•M•S PCT™
Important Biomarker for Improved Patient Management
Detects procalcitonin, a biomarker that aids in the risk assessment for progression to severe sepsis and septic shock. PCT also aids in decision making on antibiotic therapy for patients with lower respiratory tract infections (LRTI).
-
BIOMERIEUX LAB CONSULTANCY AND CHANGE MANAGEMENT SERVICES
Microbiology Expertise and Consultancy to Optimize Lab Performance
Transforming laboratories together to control costs, maximize quality, shorten time to result, and empower staff.
-
Connectivity and Data IT
Data-Driven Decision Making
Decision-making can be based on facts and figures that are easily available when needed, to measurably improve the quality, safety, efficiency, and cost of patient care.
-
VILINK®
Optimize Instrument UPTIME to Improve Laboratory Productivity
A secure solution offering remote access, proactive maintenance, and remote updates. VILINK® diagnoses, solves software and instrument issues, and supports your systems to enable laboratory productivity and efficiency improvements.
-
BIOMERIEUX LAB CONSULTANCY AND CHANGE MANAGEMENT SERVICES
Microbiology Expertise and Consultancy to Optimize Lab Performance
Transforming laboratories together to control costs, maximize quality, shorten time to result, and empower staff.
Useful Resources on Antimicrobial Stewardship
Contact Us About Our Antimicrobial Stewardship Solutions
Contact Us
References
1. How antibiotic resistance happens - centers for Disease Control. (n.d.). Available at https://www.cdc.gov/antibiotic-use/media/pdfs/how_antibiotic_resistance_happens_508.pdf Accessed on August 7, 2023.
2. The Lancet. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Available at https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(21)02724-0/. Accessed on Feb. 24, 2023.















-1?qlt=85&ts=1778522159030&dpr=off)


