VITEK® REVEAL™
Fast Antimicrobial Susceptibility Testing (AST) for Bloodstream Infections
The newest addition to our Sepsis Solution, VITEK® REVEAL™ offers fast AST results to effectively and efficiently manage bloodstream infections.
Disclaimer: Product availability varies by country. Please consult your local bioMérieux representative for product availability in your country.

- Image
- System Overview
- Services & Support
- Specs & Resources
System Overview
Optimize time to targeted therapy for patients with bloodstream infections
Within our Sepsis Solution, VITEK® REVEAL™ offers extensive antimicrobial coverage for gram-negative bloodstream infections delivering phenotypic AST results in an average of 5.5-6 hours1,2 directly from positive blood culture to better manage bloodstream infections.
Predicting antimicrobial resistance (AMR), can be more complex and time-intensive for patients with gram-negative infections. Early diagnosis and appropriate treatment make a critical difference when it comes to improving bloodstream infection management and sepsis patient outcomes. Hourly delays in antibiotic administration were associated with increased odds of hospital mortality even among patients who received antibiotics within 6 hours.3
Our comprehensive Sepsis Solution, with the addition of VITEK® REVEAL™, helps manage your patient’s antibiotic therapy, bringing you fast and reliable results to support clinical decisions for better patient care.

Data from Liu VX et al. The Timing of Early Antibiotics and Hospital Mortality in Sepsis.
Fast & accurate results directly from positive blood culture
- Real-time reporting of minimum inhibitory concentration (MIC), with first results available as early as 3 hours1
- Comprehensive and clinically relevant bug/drug combinations (covering different interpretation guidelines)
- Allows faster antimicrobial therapy modification to support antimicrobial stewardship4
Easily integrated within your lab
- Flexible solution with a modular design to fit any lab
- Access to all services from consultancy to installation by bioMérieux.
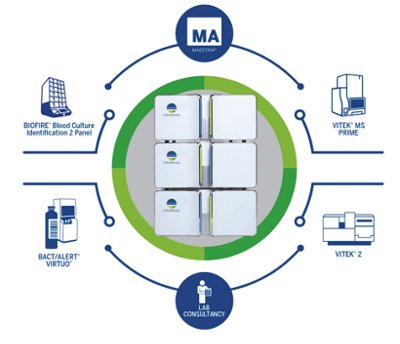
- Fully integrated sepsis solution from positive blood culture (BACT/ALERT® VIRTUO®) to identification (BIOFIRE® BCID2 Panel & VITEK® MS PRIME), AST (VITEK® 2 & VITEK® REVEAL™), and data analysis (MAESTRIA™)
References
1. BMX.1.129899 Clinical Trial Summary Report (proprietary, on file at bioMérieux).
2. Rottman M et al. Clinical evaluation of the VITEK® REVEAL™ (previously branded SPECIFIC REVEAL) Rapid AST System with Gram-negative bacteremia samples in 6 hospitals in France and England. Poster presented at: ECCMID; 2022 May; Lisbon, Portugal.
3. Liu VX et al. Am J Respir Crit Care Med. 2017. Vol 196,(7): 856–863
4. Yuceel-Timur I et al. J Antimicrob Chemother 2024; 79 Suppl 1: i26–i31 https://doi.org/10.1093/jac/dkae280.
Services & Support
VITEK® REVEAL™ is part of a comprehensive service offering from consultancy to installation and connectivity through training, verification, workflow optimization, and ongoing support.
System Services
To provide you with the highest level of customer service, training, and support we have two dedicated teams to answer your every day questions and needs:
- Local Subsidiary Teams
- Global Customer Service (GCS)
Every bioMérieux subsidiary and distributor has its own Local Customer Service to ensure support is close at hand. Our objective is to ensure you receive first class personalized service regardless of your geographical location.
These local teams can rely on global level teams, which have an international vision of the in vitro diagnostics market, for extra support when required on instruments, reagents, information technology, and service solutions.
Learning Services
bioMérieux wants to make your life easier. Login or Register to our new Resource Center website. Here you can access Quality Control Certificates (QCC), Instructions for Use (IFU), and System Manuals along with other file types.
BIOMÉRIEUX LAB CONSULTANCY
Collaborative every step of the way, we bring you customizable services that help you create the balance that makes the most of your lab talent and expertise. To really get to know your lab, we “roll up our sleeves” and work by your side to:
- Support rapid decisions
- Increase productivity
- Maximize quality
When we combine your knowledge of your lab and our expert outside perspective, we have the winning formula to enhance use of technology, improve workflow and processes, and make the most of staff skills.
Specs & Resources
Technical Specifications
VITEK® REVEAL™ Instrument
| Specification | |
|---|---|
| Width | ≤ 44 cm (17.3 in) |
| Height | ≤ 21 cm (8.3 in) |
| Depth | ≤ 72 cm (28.3 in) |
| Rear clearance | 13 cm (5.1 in) |
| Front clearance | 39 cm (15.4 in) |
| Side clearance | 13 cm (5.1 in) |
| Weight | 35 kg (77 lb) |

VITEK® REVEAL™ Sealer
| Specification | |
|---|---|
| Width | ≤ 26 cm (10.2 in) |
| Height | ≤ 45 cm (17.7 in) |
| Depth | ≤ 58 cm (28.8 in) |
| Rear Clearance | 12.7 cm (5 in) |
| Front Clearance | 21 cm (8.3 in) |
| Side Clearance | 13 cm (5.1 in) |
| Weight | 35 kg (77 lb) |

Software
| Specification | |
|---|---|
| Number of Processor Modules Connected | Up to 8 |
| Graphical User Interface | 1 Display |
| User Input Modes | Keyboard, Touch Screen, Mouse, & Barcode Reader |







