VIDAS® TBI (GFAP, UCH-L1)
RULES-OUT Intracranial Lesions in mild Traumatic Brain Injury
Fast all-in-one test kit based on blood-based brain biomarkers to predict absence of intracranial lesions in mild Traumatic Brain Injury patients and objectively assess the need for a head CT-scan.
Disclaimer: Product availability varies by country. Please consult your local bioMérieux representative for product availability in your country.


- image
- image
- Overview
- Assays
- Resources
Overview
Standard patient assessment methods for mild Traumatic Brain Injury (mTBI) can be subjective and time-consuming. Head CT-scans are frequently performed to check for intracranial lesions (ICLs). ICLs are serious but rare, so a high number of head CT-scans prove to be unnecessary. VIDAS® TBI (GFAP, UCH-L1) fills a gap in triage methods by safely ruling out ICLs and offering objective and accurate information to help determine if a head CT-scan is needed in adult patients.
- Simultaneously tests for GFAP and UCH-L1, blood-based brain biomarkers released after brain injury
- Safely rules out intracranial lesions and enables objective determination of need for head CT-scans
- Broad eligible patient population and large testing window of up to 12 hours after injury
Safely RULES-OUT Intracranial Lesions (ICL)
VIDAS® TBI (GFAP, UCH-L1) simultaneously tests glial fibrillary acidic protein (GFAP) and ubiquitin C-terminal hydrolase-L1 (UCH-L1), blood-based brain biomarkers that are released from different cell types in the hours following a brain injury. Elevation of any of these biomarkers may indicate ICL while if both are low (below specific levels), ICL can be safely ruled out. Using VIDAS® TBI (GFAP, UCH-L1) can shorten total workup time by avoiding unnecessary head CT-scans for mTBI patients while helping clinicians have confidence in their patient management decisions.
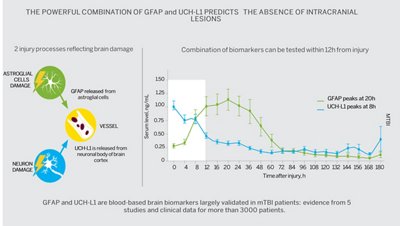
vidas_tbi_graph
Objectively determine if a head CT-scan is necessary in mTBI patients
- Wide adult patient population can benefit from this test
- Large testing window of up to 12 hours after the injury
- Easy to interpret:
- Both GFAP and UCH-L1 Negative - Can rule out the need for a head CT-scan by predicting the absence of intracranial lesions.
- GFAP and/or UCH-L1 Positive - The next step is often a head CT-scan
- High sensitivity (96.7%); High NPV (99.5%); Good specificity (41.2%)
- Avoiding unnecessary head CT-scans:
- Optimizes use of resources
- Helps reduce time spent in the ED
- Reduces patient work up time
- Reduce exposure to radiation
- Results in less than 40 minutes
- Potentially discharge patients earlier and reduce patient anxiety
VIDAS® Solutions
- Reliable and easy-to-use instruments with random access and small footprint.
- Well adapted to rapid response laboratories.
- Factory-calibrated, single-dose tests which reduce the need for additional controls.
- Short time to result.
- Reagents can be used immediately after removal from the refrigerator.
REFERENCES
Peterson B, Zhou H, Thomas KE, Daugherty J. CDC Surveillance Report 2017: Traumatic Brain Injury-related Hospitalizations and Deaths by Age Group, Sex, and Mechanism of Injury. https://www.cdc.gov/traumatic-brain-injury/
Levin H, Lancet Neurol 2015; https://www.thelancet.com/journals/laneur/article/PIIS1474-4422(15)00002-2/fulltext
Source: https://www.center-tbi.eu/patients/facts
Gil-Jardiné C, et al. MANAGEMENT OF PATIENTS SUFFERING FROM MILD TRAUMATIC BRAIN INJURY 2023. Anaesth Crit Care Pain Med. 2023 Jun 5:101260.
Bazarian J, et al. Serum GFAP and UCH-L1 for prediction of absence of intracranial injuries on head CT (ALERT-TBI): a multicentre observational study. Lancet Neurol. 2018;17:782-789.
Papa L, Brophy GM, Welch RD, et al. Time Course and Diagnostic Accuracy of Glial and Neuronal Blood Biomarkers GFAP and UCH-L1 in a Large Cohort of Trauma Patients With and Without Mild Traumatic Brain Injury. JAMA Neurol. 2016 May 1;73(5):551-60
VIDAS® TBI Package Insert (or IFU) last version as per IVDR approval.
Assays
| Assays | Reference | Tests per Kit | Code | Time to Result | Decisional Cut-Offs |
|---|---|---|---|---|---|
| VIDAS® TBI (GFAP, UCH-L1) | 423615-30 | 30 tests | GFAP UCH 1 patient test = 1 GFAP + 1 UCH-L1 | 39 min | GFAP= 22pg/mL UCH-L1 = 327 pg/mL |
BECAUSE IT MAKES SENSE ON VIDAS®






