News
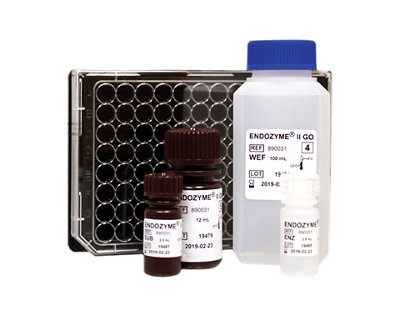
bioMérieux, a world leader in the field of in vitro diagnostics, announces the launch of BIOFIRE® SPOTFIRE® for pharmaceutical industries, where this innovative molecular testing system is designed to transform quality control. Building on the proven success of the BIOFIRE® FILMARRAY® platform, already adopted by leading global pharmaceutical companies, BIOFIRE® SPOTFIRE® introduces a simple, small and smart approach to mycoplasma detection, helping biopharmaceutical manufacturers strengthen quality control, accelerate decision-making, and protect patients.

Swipe to discover more
-
Global Agreement Announced at G7 One Health Summit to Accelerate Access to Diagnostics and Responses to Rising Health Threats
A coalition of more than 20 international partners announced the Global One Health Diagnostics Access Compact (GO-Dx) during the G7 One Health Summit in Lyon, France. This new initiative serves as a catalyst for international leaders across different sectors to advance progress toward expanding diagnostics access, enabling early detection and better surveillance, and improving response against existing and emerging threats across human, animal, and environmental health. By aligning efforts, this group aims to strengthen global preparedness through science based One Health solutions. -
Always consider sepsis in the context of infection.
Patient associations across the globe are actively engaged in promoting this critical message to both healthcare professionals and the broader public. We met with Dr Rafael Moraes from ILAS, Melissa Mead from The UK Sepsis Trust and Thomas Heymann from Sepsis Alliance to learn more about their actions. -
Predicting Sepsis-Related Shock Earlier: How AI Could Transform Critical Care
In the ICU, minutes count — particularly for patients with sepsis, where a sudden drop in blood pressure can signal a dangerous turn. If care teams can see that change coming, even by a couple of hours, they can prepare, personalize treatment, and potentially prevent a crisis. That’s where interpretable, clinically grounded AI can help — turning routine hospital data into early warning signs.
Enabling Decision-Making
- ANTIMICROBIAL STEWARDSHIP
- SEPSIS
- FOOD SAFETY & QUALITY
- PHARMA QUALITY CONTROL
- COVID-19
- ANTIMICROBIAL STEWARDSHIP
- SEPSIS
- FOOD SAFETY & QUALITY
- PHARMA QUALITY CONTROL
- COVID-19














?qlt=85&ts=1670238196189&dpr=off)