BIOFIRE® FILMARRAY® 2.0 EZ Configuration
The BIOFIRE 2.0 EZ Configuration facilitates rapid near-patient molecular diagnostic testing with results in about 45 minutes.


- Product Overview
- Service & Support
- Resources
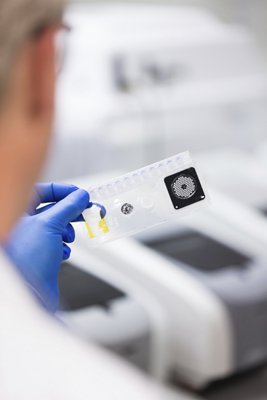
Product Overview
The BIOFIRE FILMARRAY 2.0 EZ Configuration
- Designed for use with the BIOFIRE® Respiratory 2.1-EZ (RP2.1-EZ) Panel (EUA)* in CLIA-waived testing sites, including clinics and physician offices
- Enables decentralized molecular testing throughout a provider network
- Features a simplified and intuitive interface and results report
CLIA Certificate of Waiver required to perform testing.
Not available outside of the U.S.

How It Works
The BIOFIRE 2.0 EZ Configuration offers ease-of-use with just two minutes of hands-on time and results in about 45 minutes. Training is minimal and the software walks you through the entire molecular testing process.
Fast, Easy, Comprehensive
- Increased Speed and Cost Efficiency: The BIOFIRE 2.0 EZ Configuration delivers actionable answers in about 45 minutes. Faster results may lead to fewer downstream dignostic tests, may improve antimirobial stewardship, and may support better infection control—all of which may contribute to better patient satisfaction.
- User-Friendly Multiplex PCR: Any tech on any shift can use the BIOFIRE 2.0 EZ Configuration, which requires minimal training and hands-on time to help optimize laboratory productivity 24/7.
- The Most Comprehensive Target Menu on the Market: One test on the BIOFIRE 2.0 EZ Configuration simultaneously targets the most likely causes of respiratory infection, empowering clinicians to choose the right test, the first time. Results are pathogen-specific, clinically relevant, and can lead to more informed patient management decisions.

Service & Support
With the urgent nature of patient and community care, we take pride in addressing all concerns quickly and accurately. bioMérieux is dedicated to providing world class customer support 24 hours a day, 7 days a week, 365 days a year.
For assistance please contact our customer technical support team at:
- Email: biofiresupport@biomerieux.com
- Phone: 801-736-6354 option 5
- Toll Free: 1-800-735-6544
To order, contact your regional sales representative or use the following contact information:
- Phone: +1-801-736-6354 x 1502
- Fax: +1-801-588-0507
- Email: CSCFax@biomerieux.com
- International Sales: +1-801-736-6354 x 1536
- Military Sales: 1-801-262-3592
Resources
Technical & Supporting Documents
Videos
Be a Diagnostic Genius with the BIOFIRE 2.0 EZ Configuration
BIOFIRE System - How it Works
BIOFIRE System - How it Works
*This product has not been FDA cleared or approved, but has been authorized for emergency use by FDA under an EUA for use by authorized laboratories; This product has been authorized only for the detection and differentiation of nucleic acid of SARS-CoV-2 from multiple respiratory viral and bacterial organisms; and, The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.