BIOFIRE® FILMARRAY® TORCH System
The BIOFIRE TORCH System uses multiplex PCR technology to simultaneously test for a comprehensive grouping of targets in about an hour.



- Product Overview
- Service & Support
- Resources
Product Overview
The BIOFIRE TORCH System is the latest advancement in molecular infectious disease diagnostics.
- Compatible with six syndromic BIOFIRE® FILMARRAY® Panels.
- Reduced footprint provides up to six times the throughput per square foot of laboratory bench space
- Scalable configuration allows customized throughput
- Simplified workflow features a touchscreen interface and integrated barcode scanner
- Data management automation includes LIS connectivity and random-access operation

The BIOFIRE TORCH System – How It Works
The BIOFIRE System offers unmatched ease-of-use with just two minutes of hands-on time and results in about an hour. Training is minimal and the software walks you through the entire molecular testing process.
It takes just three easy steps to get faster disease diagnostic results with our multiplex assay technology.
- Setup—inject hydration solution and an unprocessed sample into pouch
- Test—insert pouch into the BIOFIRE System instrument and start the run
- Get Result—software reports whether each target is detected in the sample
The BIOFIRE® FILMARRAY® Pouch is a multiplex PCR assay that stores all the necessary reagents for sample preparation, reverse transcription PCR, PCR, and detection in a freeze-dried format. During a test run, the BIOFIRE System extracts and purifies all nucleic acids from the unprocessed sample. Next, it performs nested multiplex PCR in two stages. The first stage includes a single, large volume, multiplexed reaction. The second stage includes individual, single-plex reactions to detect the products from the first stage.
Using endpoint melting curve data, the BIOFIRE System software automatically analyzes the results for each target on the panel. When the run is complete, the software reports whether each pathogen is detected in the sample. This information is printed in an automated response at the end of the test run.

Fast, Easy, Comprehensive
- Increased Speed and Cost Efficiency: The BIOFIRE System delivers actionable answers in about an hour. Faster results may lead to fewer downstream diagnostic tests, may improve antimicrobial stewardship, and may support better infection control—all of which may contribute to better patient satisfaction.
- User-Friendly Multiplex PCR: Any tech on any shift can use the BIOFIRE System, which requires minimal training and hands-on time to help optimize laboratory productivity 24/7.
- The Most Comprehensive Target Menu on the Market: One test on the BIOFIRE System simultaneously targets the most likely causes of infection, empowering clinicians to choose the right test, the first time. Results are pathogen-specific, clinically relevant, and can lead to more informed patient management decisions.

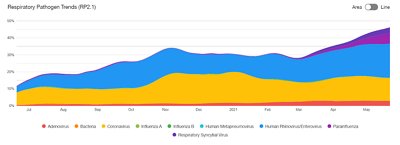
BIOFIRE® Syndromic Trends
BIOFIRE Syndromic Trends provides visual insight into local and regional pathogen circulation trends on-demand. This software feature, available to bioMérieux customers, makes it possible to monitor infectious disease data in near real-time while preserving patient privacy.
Product availability varies by country. Consult your bioMérieux representative.
Service & Support
With the urgent nature of patient and community care, we take pride in addressing all concerns quickly and accurately. bioMérieux is dedicated to providing world class customer support 24 hours a day, 7 days a week, 365 days a year.
For assistance please contact our customer technical support team at:
- Email: biofiresupport@biomerieux.com
- Phone: 801-736-6354 option 5
- Toll Free: 1-800-735-6544
To order, contact your regional sales representative or use the following contact information:
- Phone: +1-801-736-6354 x 1502
- Fax: +1-801-588-0507
- Email: CSCFax@biomerieux.com
- International Sales: +1-801-736-6354 x 1536
- Military Sales: 1-801-262-3592
Resources
Technical & Supporting Documents
Videos
Watch How the BIOFIRE TORCH System can help with your syndromic infectious disease testing needs.
BIOFIRE Torch System